Professor Timothy Jackson named May 2022 Sutton Family Research Impact Award recipient

The Department of Chemistry congratulates Professor Timothy Jackson on receiving the May 2022 Sutton Family Research Impact Award!
The Sutton Award is a monthly competition among chemistry faculty. Every month, the Chemistry Department Chair and Associate Chairs review the peer-reviewed papers published by chemistry faculty from the three previous months to select a winner. The recipient receives a $500 cash prize and is featured on the departmental website.
For a full list of winners, visit our Sutton Family Research Impact Award webpage.
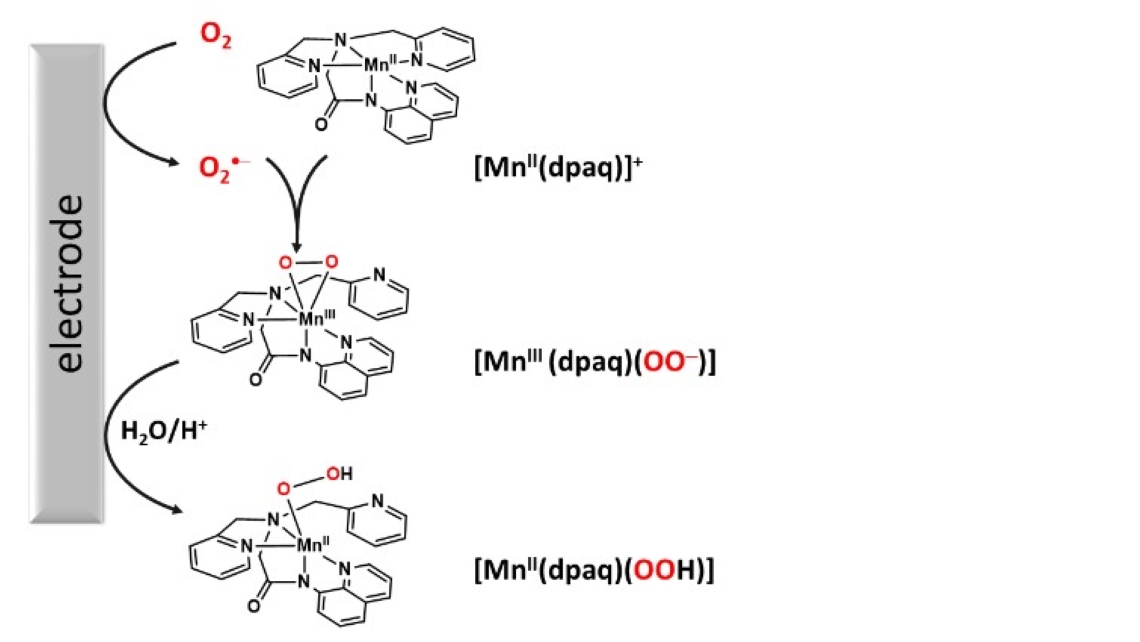
Electrochemical Formation and Reactivity of a Mn-Peroxo Complex Bearing an Amido N5 Ligand
By Dr. Allyssa A. Massie, Dr. Nikolaos Kostopoulos, Elizabeth N. Grotemeyer, Dr. Jean-Marc Noël, Dr. Timothy A. Jackson*, and Dr. Elodie Anxolabéhère-Mallart
Published in: ChemElectroChem 2022, 9, e202200112.
One of the hallmark reactions of metals in biological systems is the reduction of the oxygen molecule. In this reaction, a metal center binds the oxygen molecule and cofactors deliver electrons for the reduction reaction. This reaction is critical for biological processes ranging from aerobic respiration to the biosynthesis of hormones. Mimicking this process in synthetic chemistry has remained a challenge. A recent experimental-computational study revealed how electrochemical methods and synthetic manganese complexes can be combined to help control this oxygen reduction reaction. This project began through an international supplement to a National Science Foundation grant awarded to Prof. Tim Jackson. Allyssa Massie (KU Ph.D. 2018) spent two months in Paris performing electrochemical experiments with Dr. Elodie Anxolabéhère-Mallart, the Director of the Laboratoire d’Electrochimie Moléculaire at the Université Paris Diderot. Liz Grotemeyer, a current graduate student in the Jackson group, performed computational studies to aid in understanding the experimental data. Collectively, this work revealed that electrochemical methods can be used to control the delivery of electrons to a manganese-oxygen complex, mimicking the delivery of electrons by cofactors in biology. This work also demonstrated that these electrochemical methods can be used generate new manganese-oxygen intermediates that had been inaccessible through other routes.