Professor Ward Thompson named August 2023 Sutton Family Research Impact Award recipient

The Department of Chemistry congratulates Professor Ward Thompson on receiving the August 2023 Sutton Family Research Impact Award!
The Sutton Award is a monthly competition among chemistry faculty. Every month, the Chemistry Department Chair and Associate Chairs review the peer-reviewed papers published by chemistry faculty from the three previous months to select a winner. The recipient receives a $500 cash prize and is featured on the departmental website.
For a full list of winners, visit our Sutton Family Research Impact Award webpage.
Relation between the Hydrated Electron Solvation Structure and Its Partial Molar Volume
By Pauf Neupane, David M. Bartels,* and Ward H. Thompson,*
Published in Journal of Physical Chemistry B, 2023, 127, 5941-5947
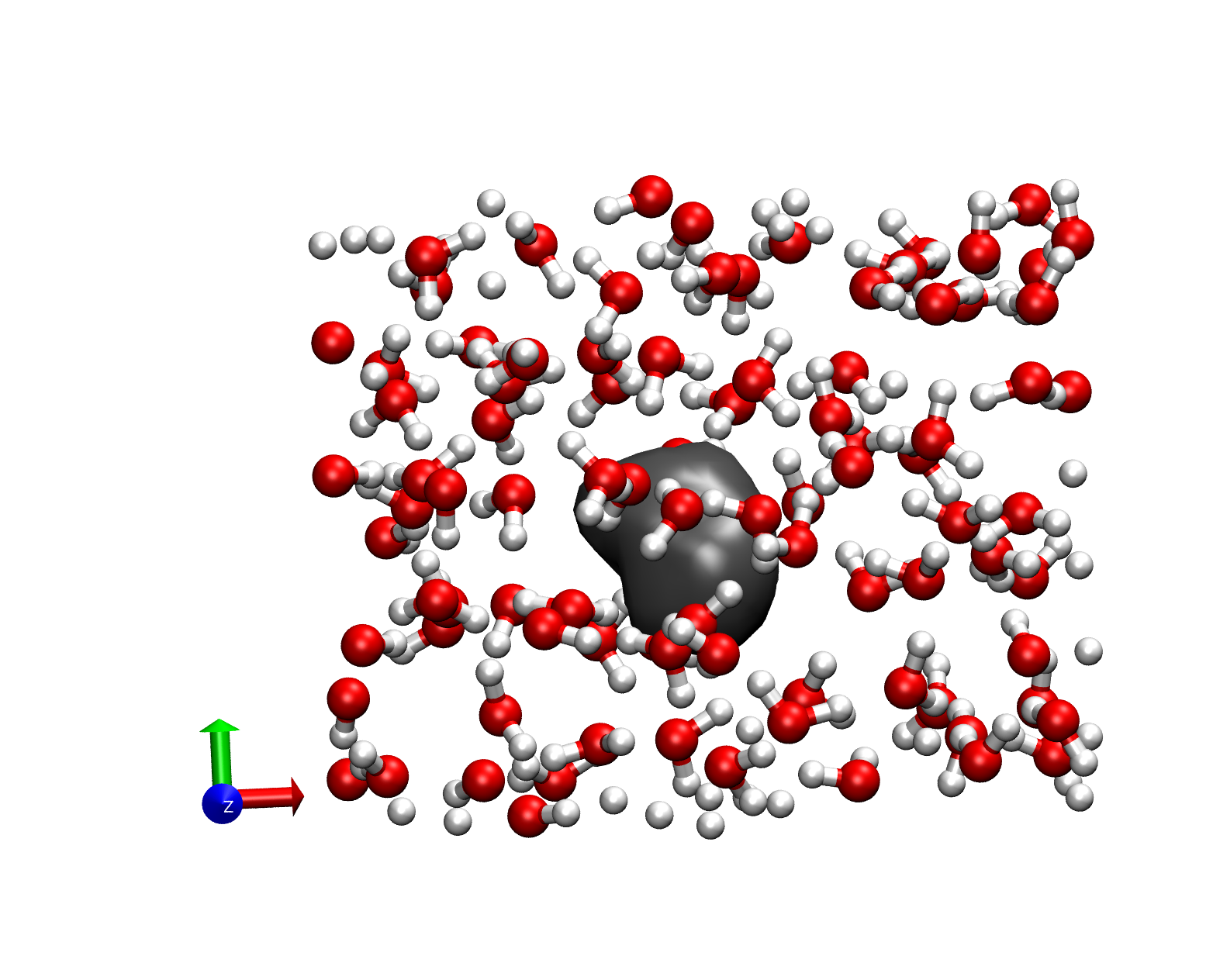
Hydrated electrons are formed when radiation hits water and detaches an electron. Because a water molecule cannot form a stable anion, the hydrated electron exists unattached to any single water molecule (see figure which shows the hydrated electron in gray). In this way it is like a nucleus-less atom and, as one might imagine for an electron without a nucleus, it is a powerful reducing agent. This makes it an important part of mechanisms for corrosion in nuclear reactors and radiation-induced damage in DNA. Despite intense study over many decades there are still many open questions about the hydrated electron, including about its basic structure. Indeed, the past 15 years have seen an extensive discussion in the literature about whether the electron occupies a cavity within the water or not. The cavity picture has emerged as the consensus description, but among the issue raised in this debate is the interpretation of the hydrated electron partial molar volume. This measures the change in volume of water when a hydrated electron is added, i.e., how much, and in which way, does the hydrated electron change the volume of the solution.
In this paper, the Thompson group showed how the partial molar volume can be readily calculated from structural data extracted from molecular simulations. The work was carried out by Dr. Pauf Neupane, a postdoctoral associate in the Thompson group, who executed simulations in which the quantum Schrödinger equation is solved at every timestep in a classical molecular dynamics simulation of the water molecules. The density of the water around the hydrated electron is then calculated as a function of distance and this is used to determine the partial molar volume using what is known as Kirkwood-Buff theory. An important advantage of this approach is that it makes clear the relationship between the water structure around the hydrated electron and the partial molar volume. Dr. Neupane examined multiple models that have been proposed for describing the hydrated electron. His results confirmed that what is generally considered the most accurate of these models gives the wrong sign of the partial molar volume. Namely, it predicts the solution contracts when a hydrated electron is added. However, measurements by Prof. David Bartels, a collaborator in this work, previously determined the partial molar volume to be positive, i.e., addition of the hydrated electron leads to expansion of the solution. More importantly, Dr. Neupane could determine the contribution to the partial molar volume from the different parts of the system, e.g., the size of the cavity formed by the hydrated electron and the structuring of the water in the first few solvation shells. (The first solvation shell is made up of waters next to the hydrated electron, the second of water molecules next to those, and so on.) These results showed that, despite some assumptions to the contrary in the literature, the partial molar volume cannot be used to infer the cavity size (or even if the electron resides in a cavity). Rather, the structuring of water molecules, even in the second and third solvation shells, has a large effect. This gives a more subtle, and more interesting, interpretation of the hydrated electron partial molar volume.