Prof. Misha Barybin named November 2023 Sutton Family Research Impact Award recipient
The Department of Chemistry congratulates Professor Mikhail "Misha" Barybin on receiving the November 2023 Sutton Family Research Impact Award!
The Sutton Award is a monthly competition among chemistry faculty. Every month, the Chemistry Department Chair and Associate Chairs review the peer-reviewed papers published by chemistry faculty from the three previous months to select a winner. The recipient receives a $500 cash prize and is featured on the departmental website.
For a full list of winners, visit our Sutton Family Research Impact Award webpage.
Homoleptic complexes of isocyano- and diisocyanobiazulenes with a 12-electron, ligand-based redox capacity
By Patrick T. Connelly, Jason C. Applegate, David A. Maldonado, Monisola K. Okeowo, Wade C. Henke, Allen G. Oliver, Cindy L. Berrie, and Mikhail V. Barybin
Published in Dalton Trans., 2023, 52, 11419-11426
Polycyclic aromatic hydrocarbons (PAH’s) and their derivatives have been at the core of impressive innovations at the interface of chemistry, physics, materials science, biotechnology, and engineering. A diverse range of benzenoid PAH scaffolds (i.e., those involving 6-membered benzene rings) is ubiquitously accessible, yet their widespread use constitutes a “double-edged sword” due to the well-documented serious biological and environmental toxicity exerted by many PAH’s. Being a completely nonbenzenoid PAH, the azulenic motif, along with its higher homologues, deserves special attention as it represents a more general type of electronic aromaticity compared to the alternant (e.g., benzenoid) systems, where π-electrons are dispersed evenly over even-membered rings. The “parent” compound – azulene – is a polar hydrocarbon featuring an edge sharing combination of 5- and 7-membered carbon rings. Notably, the toxicology, carcinogenicity, and mutagenicity profiles of azulene itself are far more favorable compared to those of its benzenoid isomer naphthalene. Due to their unusual physicochemical characteristics, azulenes and polyazulenes constitute attractive redox addressable, optoelectronic, and conductive molecular building blocks.
Electrical energy storage within organic molecules capable of rapidly and reversibly exchanging electrons through low-voltage redox activity has been attracting increasing interest as an alternative to metal-based electron reservoirs. As the centerpiece of their recent collaborative article in Dalton Transactions, the research teams of Professors Misha Barybin and Cindy Berrie introduced mono- and heptanuclear chromium complexes that feature 12 azulenic units per molecule (Figure 1). These well-defined organometallic molecular architectures exhibit reversible 12-electron reduction capacity concentrated on the organic units and are distinctly different from metal-based organometallic electron reservoirs. Notably, the mononuclear and heptanuclear chromium “nanocomplexes” illustrated in Figure 1 have overall reversible redox capacities of 15 and 21 electrons, respectively. The authors also presented ultraviolet-visible and vibrational spectroscopic evidence suggesting that their diisocyanobiazulenic π-linker mediates readily quantifiable electronic communication at an impressive 2 nm distance not only in electron-rich polynuclear chromium complexes but also in a gold/chromium heterobimetallic system involving an organochromium molecular monolayer film self-assembled on a metallic gold surface.
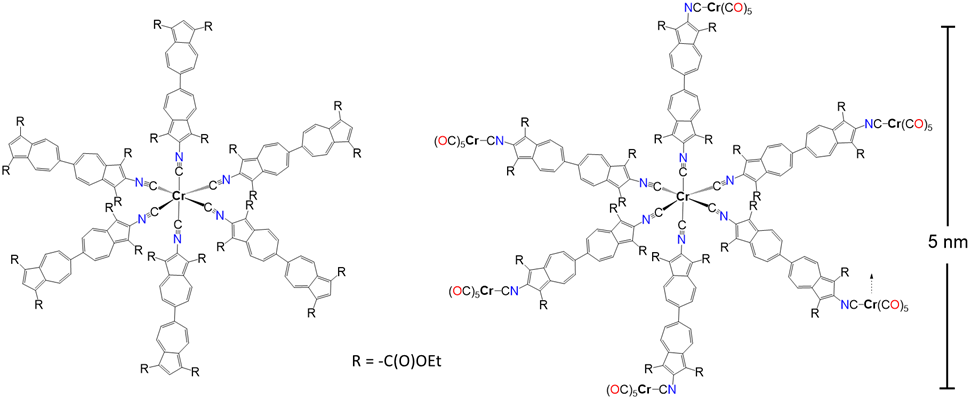
Figure 1. Mononuclear (left) and heptanuclear (right) organochromium nanocomplexes with 15- and 21-electron reversible redox capacities.

Figure 2. Patrick Connelly, a doctoral candidate in Prof. Barybin’s research group and co-first author of the article in Dalton Transactions.