Minae Mure named December 2022 Sutton Family Research Impact Award recipient

The Department of Chemistry congratulates Associate Professor Minae Mure on receiving the December 2022 Sutton Family Research Impact Award! Her winning paper was published in International Journal of Molecular Science and is titled, "Insight into the Spatial Arrangement of the Lysine Tyrosylquinone and Cu2+ in the Active Site of Lysyl Oxidase-like 2."
The Sutton Award is a monthly competition among chemistry faculty. Every month, the Chemistry Department Chair and Associate Chairs review the peer-reviewed papers published by chemistry faculty from the three previous months to select a winner. The recipient receives a $500 cash prize and is featured on the departmental website.
For a full list of winners, visit our Sutton Family Research Impact Award webpage.
Insight into the Spatial Arrangement of the Lysine Tyrosylquinone and Cu2+ in the Active Site of Lysyl Oxidase-like 2
By Alex A. Meier, Hee-Jung Moon, Sinan Sabuncu, Priya Singh, Trey A. Ronnebaum, Siyu Ou, Justin T. Douglas, Timothy A. Jackson, Pierre Moënne-Loccoz, and Minae Mure
Published in the International Journal of Molecular Sciences (Vol 23) 13966 (2022)
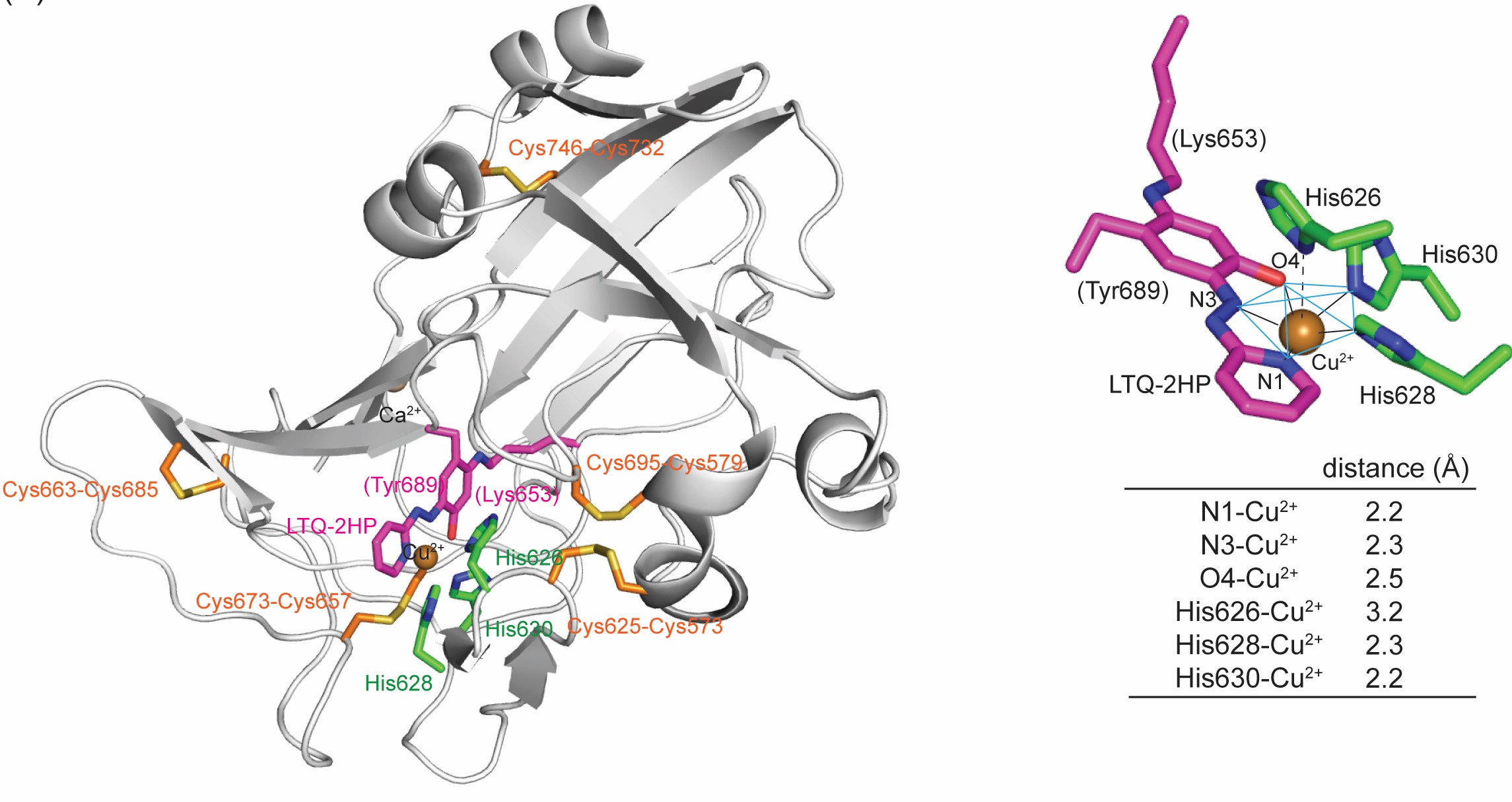
Prof. Minae Mure recently published an article in the International Journal of Molecular Science (Vol 23) 13966 (2022) on assessing the spatial arrangement of the lysine tyrosylquinone (LTQ) cofactor and the active site Cu2+ in lysyl oxidase-like 2 (LOXL2). LOXL2 is Cu2+-dependent amine oxidase that is involved in tumor metastasis/invasion and organ fibrosis and is recognized as a promising therapeutic target against these diseases. Currently, there is no structural information of the catalytically-active LOXL2, and structure-based drug design has been severely hampered. In this study, the spatial arrangement of the LTQ cofactor and the active site Cu2+ was assessed for the first time, and they reside within 2.9 Å to each other. The Mure lab has overcome the hurdle in obtaining recombinant LOXL2 with high catalytic activity and subjected the protein sample for biophysical and biochemical characterization. The LTQ cofactor in the recombinant LOXL2 was quantitated by UV-vis spectroscopic titration with 2-hydrazinopyridine (2HP) to be a nearly stoichiometric amount. The origin of the raspberry pink color of the 2HP-modified LOXL2 turns out to be LTQ-2HP (the LTQ cofactor covalently modified with 2HP) ligated to the active site Cu2+. The identification of LTQ-2HP in LOXL2 was achieved by combination of protein chemistry and model chemistry (organic chemistry + analytical chemistry + inorganic chemistry) in collaboration with Drs. Timothy Jackson and Justin Douglas (KU Chemistry) on EPR spectroscopy and Dr. Pierre Moënne-Loccoz (Oregon Health Science University) on resonance Raman spectroscopy. The results from this study have enabled the Mure lab to generate the first active site structure of LOXL2 containing the LTQ cofactor and Cu2+ in collaboration with Dr. Kryzsztof Kuczera (KU Chemistry) by 3D-modeling [A 3D-predicted structure of the Amine Oxidase Domain of Lysyl Oxidase-like 2, Int. J. Mol. Sci. 2022, 23, 13385]. In collaboration with Dr. Kuczera, the Mure lab has just begun docking study of small molecule inhibitors that they have identified from high throughput screening (HTS) of 25,000 small molecules at KU-HTS laboratory (Dr. Anuradha Roy).