James Blakemore named June 2023 Sutton Family Research Impact Award recipient

The Department of Chemistry congratulates Associate Professor James Blakemore on receiving the June 2023 Sutton Family Research Impact Award!
The Sutton Award is a monthly competition among chemistry faculty. Every month, the Chemistry Department Chair and Associate Chairs review the peer-reviewed papers published by chemistry faculty from the three previous months to select a winner. The recipient receives a $500 cash prize and is featured on the departmental website.
For a full list of winners, visit our Sutton Family Research Impact Award webpage.
Mechanistic roles of metal- and ligand-protonated species in hydrogen evolution with [Cp*Rh] complexes
By Wade C. Henke, Yun Peng, Alex A. Meier, and James D. Blakemore
Published in PNAS 120 (21) e2217189120
Hydrogen is often discussed as a “fuel of the future.” Indeed, fuel cell cars and other devices that can run on hydrogen are becoming more available. One important issue in use of hydrogen as a fuel, however, is development of technologies for generation of hydrogen that are sustainable. On the one hand, much of the hydrogen that is used in our society at present is generated by steam reforming of methane, a fossil fuel. Thus, even though the hydrogen itself is thought of as a clean fuel, if methane is used as the source of hydrogen, it is not such a clean fuel because its generation results in significant carbon dioxide by-product. On the other hand, there are great efforts underway to develop electrocatalytic approaches and electrolyzers for generation of hydrogen from water with only gaseous oxygen as the co-generated byproduct. This “green” hydrogen is cleaner, since there is not release of carbon dioxide from the process used to make it.
Surprisingly, however, the precise chemical steps involved in hydrogen generation are not well understood for most catalysts. Even platinum, perhaps the most effective catalyst for hydrogen evolution that is known, doesn’t have an established, well-defined mechanism of action. Considering this challenge, chemists are working on molecular model complexes that can be studied to gain insights into the mechanisms which underpin generation of clean hydrogen fuel from water. Inorganic chemistry has an outsize role to play in such studies, as most catalysts use metals to orchestrate the individual bond breaking and forming reactions that lead to stitching together the H–H molecule.
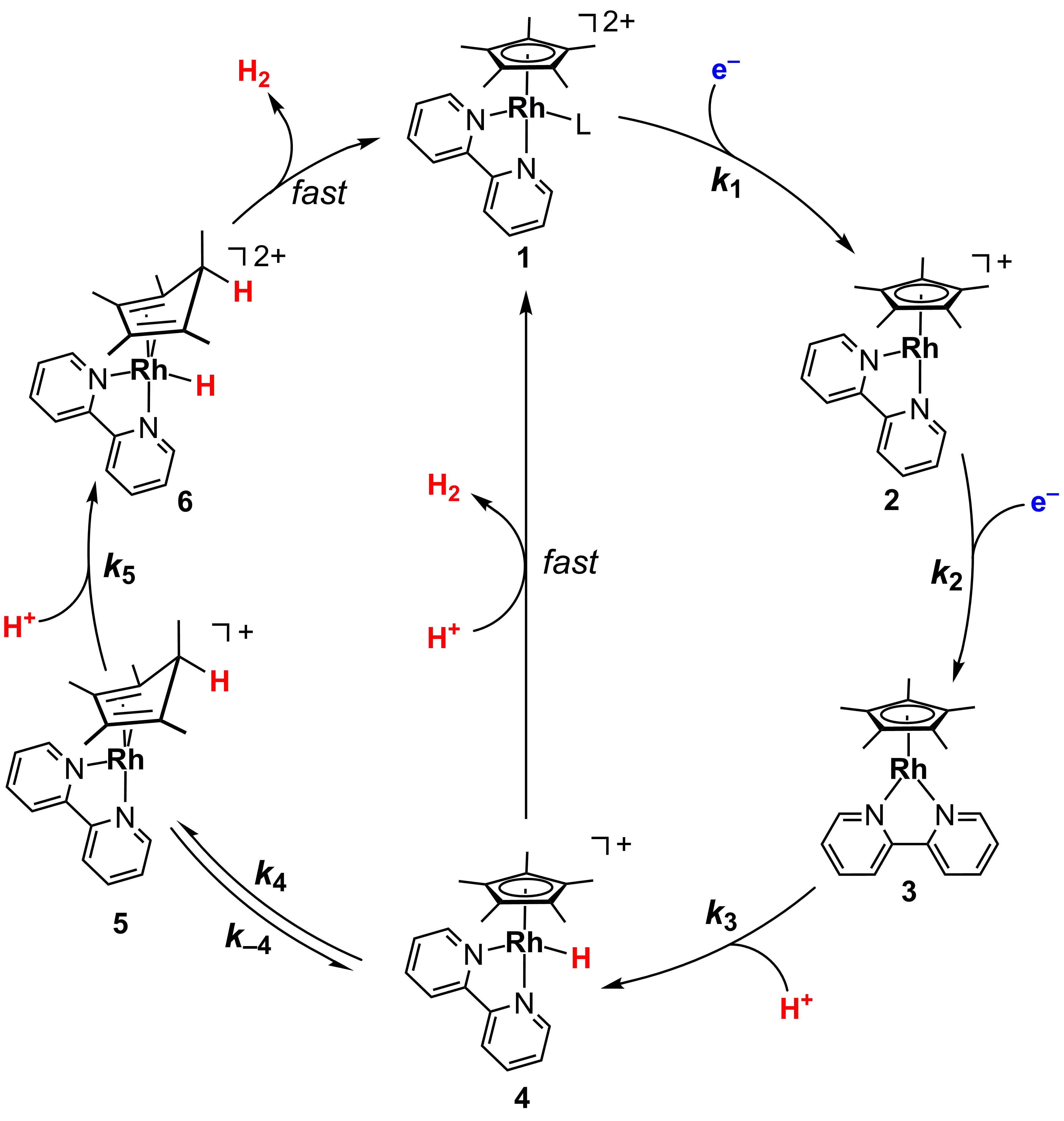
In our group’s recent paper, we partnered with Brookhaven National Laboratory to carry out mechanistic studies on a class of hydrogen-evolving catalysts that are built around a rhodium metal center. Rhodium is a very useful but rare element located in the second row of the transition metal series of the periodic table, and is perhaps best known as one of the components found in catalytic converters in cars. In our complexes, rhodium serves along with a set of organic ligands as a catalyst for generation of hydrogen. In particular, our complexes feature the pentamethylcyclopentadienyl ligand, a well-known ligand in organometallic chemistry that is commonly abbreviated as Cp*.
The chemistry at the heart of our investigation regards unusual involvement of the Cp* ligand in handling the reactions that bring together the two protons (H+) and electrons (e–) that ultimately makeup the product hydrogen, H–H. In 2016, the first evidence appeared that the Cp* ligand was involved in holding one H+ in an intermediate—this species is fundamentally different than the analogous sort of intermediate which would be more commonly involved, a rhodium hydride complex in which the H+ is directly bonded to the metal center. The chemistry of this system, and studies of the interconversion of such hydrides and protonated ligand complexes, occupied a great deal of the Blakemore Group’s research efforts between 2016 and 2022. This most recent study in Proc. Nat. Acad. Sci. unpacked the elementary reaction steps involved in the transfer of e– and H+ to the rhodium catalyst, as well as the reactions that lead to bringing them all together in the product.
The work was carried out both at KU and at Brookhaven National Laboratory. On the KU side, we prepared all the metal complexes and starting materials used in the work, as well as completed kinetic studies using stopped-flow UV-visible spectroscopy. This part of the work took advantage of the capabilities in the Blakemore Laboratory in air- and moisture-free chemistry, as well as spectroscopy. Former graduate students Wade Henke and Yun Peng in the Blakemore Group took the lead on this part of the work. At Brookhaven, our collaborators David Grills, Dmitry Polyansky and Etsuko Fujita led very specialized spectroscopic experiments using both pulse radiolysis and time-resolved UV-visible and infrared spectroscopies. The joint work represents a powerful combination of expertise that enabled study of a very unusual catalytic system using world-class, specialized techniques.
The link to the paper, “Mechanistic roles of metal- and ligand-protonated species in hydrogen evolution with [Cp*Rh] complexes” is here: Mechanistic roles of metal- and ligand-protonated species in hydrogen evolution with [Cp*Rh] complexes
This work was also featured by the KU news service, https://news.ku.edu/2023/05/16/scientists-reveal-breakthrough-could-lead-cleaner-hydrogen-energy, and Brookhaven National Laboratory also issued a news release about the work, https://www.bnl.gov/newsroom/news.php?a=121150.
The photo shows the current members of the Blakemore Group, as of July 2023.
Speaking about his group’s rhodium project broadly, Prof. Blakemore stated: “We learned a great deal about conducting redox chemistry and catalysis through our work on [Cp*Rh] complexes. The project provided new insights regarding H+ and e– management during hydrogen evolution, but more importantly, provided substantial new information about the underappreciated but potential quite useful phenomenon of proton-hydride tautomerism. Perhaps best of all, however, the project inspired several generations of both graduate students and undergraduate researchers in my group to pursue studies of inorganic and organometallic chemistry, initiating new career opportunities and illuminating quite exciting paths. Other research groups are becoming more interested in Cp* involvement in catalysis and new types of tautomerism—the field is picking up!"
About the future of this chemistry in his group, he added: “My group is still quite interested in understanding H+ and e– movement during redox reactions, and are now applying the skills honed in the rhodium work to studies of the heavy elements, particularly the early actinides thorium and uranium. We are also continuing the rhodium work, at present in collaboration with Prof. Caricato’s group. Like so many stories in science, we continue to find new, unexpected, and attractive avenues for further investigations!”