Brian Laird named September 2022 Sutton Award recipient

The Department of Chemistry congratulates Professor Brian Laird on receiving the September 2022 Sutton Family Research Impact Award!
The Sutton Award is a monthly competition among chemistry faculty. Every month, the Chemistry Department Chair and Associate Chairs review the peer-reviewed papers published by chemistry faculty from the three previous months to select a winner. The recipient receives a $500 cash prize and is featured on the departmental website.
For a full list of winners, visit our Sutton Family Research Impact Award webpage.
Inside and out: Surface thermodynamics from positive to negative curvature
By Seth C. Martin, Hendrik Hansen-Goos, Roland Roth and Brian B. Laird
Published in: J. Chem. Phys. 157, 054702 (2022).
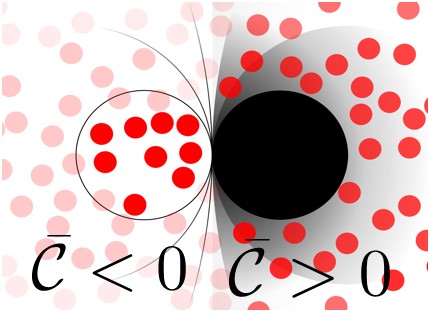
The solid-liquid interfacial free energy γ, which represents the energy cost to form an interface between a solid and a liquid phase, plays a fundamental role in the understanding of a broad array of technologically important phenomena, such as crystal nucleation and growth, nanoparticle shape and stability, as well as the wetting of surfaces by liquids – for example, the beading up of water on a freshly waxed car occurs because the presence of the wax on the surface increases γ, causing the surface to be less favorable to wetting by water than the unwaxed surface. Most fundamental studies of γ for solid-liquid interfaces involve fluids at flat surfaces; however, two interfacial systems in which the curvature dependence of γ becomes important are (a) a nanoscale solid particle immersed in a fluid and (b) a fluid confined within a nanoscale container. One major open question in surface thermodynamics is the degree to which these two seemingly very different systems can be described within a single theory. In this work, we answer this question using a model simple 2d fluid of hard disks – disks whose only interactions are elastic collisions. We then consider γ of this fluid at a hard circular nanoparticle of radius R, as well as that for the same fluid contained within a hard circular container of the same radius. The mathematical curvature